ORIGINAL ARTICLE
Three distinctive Preussia (Sporormiaceae) from photosynthetic stems of Ephedra trifurca (Ephedraceae, Gnetophyta) in southeastern Arizona, USA
1
Department of Ecology and Evolutionary Biology, The University of
Arizona, Tucson, AZ 85721, USA
2
School of Plant Sciences, The University of Arizona, Tucson, AZ
85721, USA
3
Facultad de Ciencias, Universidad Nacional Autónoma de México,
Ciudad Universitaria, CDMX, México
Online publication date: 2022-12-28
Publication date: 2022-12-28
Plant and Fungal Systematics 2022; 67(2): 63-74
KEYWORDS
ABSTRACT
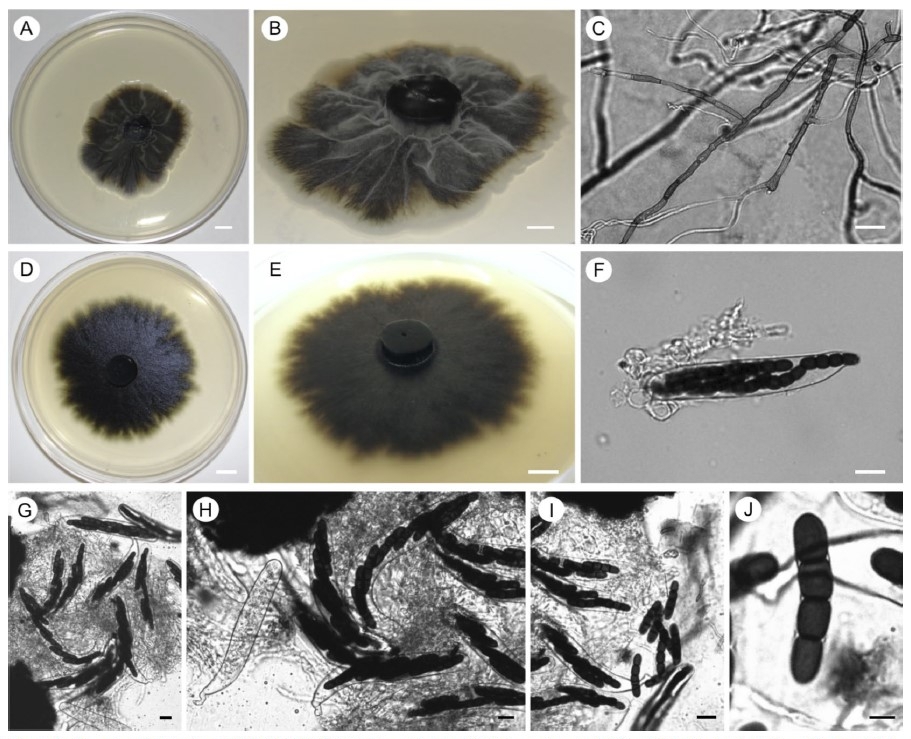
A survey of the diversity and community composition of culturable endophytic
fungi associated with photosynthetic stems of the gnetophyte Ephedra trifurca (Ephedraceae)
in southeastern Arizona, USA, yielded numerous isolates that are consistent morphologically
with members of the genus Preussia (Sporormiaceae, Pleosporales, Dothideomycetes, Ascomycota).
Preussia (including species until recently classified within Sporormiella) include
dung-, soil-, and plant-inhabiting strains with considerable diversity worldwide. We used
morphological evidence and phylogenetic analyses (nuclear ribosomal internal transcribed
spacers and 5.8s gene, the adjacent D1-D2 region of the nuclear ribosomal large subunit,
and for a subset of strains, the translation elongation factor 1-alpha, EF1-a) to identify
distinctive members of the Preussia intermedia clade occurring as endophytes in E. trifurca.
These include P. arizonica sp. nov., which also occurs as an endophyte in other plants of
the region, and P. elegans sp. nov., which has been found only in E. trifurca to date. We
also propose Preussia mariae sp. nov., allied phylogenetically with Preussia lignicola but
distinguishable on the basis of morphology and EF1-a data. Our analyses illustrate the
potential for several currently recognized species of Preussia to represent species complexes
that should be resolved by analyses of additional loci and by further sampling of
endophytes, which may provide an ecological connection among strains occurring within
living plant tissues and as coprophilous or soil-inhabiting fungi. More broadly, our work
expands the known geographic scope, host use, and diversity of Preussia, especially in arid
lands. In conjunction with previous work, our study also provides the basis for hypotheses
regarding secondary metabolites of the newly described species.
FUNDING
Financial support for this work was provided by the College of
Agriculture and Life Sciences at the University of Arizona.
REFERENCES (43)
1.
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. 1990. Basic local alignment search tool. Journal of Molecular Biology 215: 403–410. https://doi.org/10.1016/S0022-....
2.
Araldi-Brondolo, S. J., Spraker, J., Shaffer, J. P., Woytenko, E. H., Baltrus, D. A., Gallery, R. E. & Arnold, A. E. 2017. Bacterial endosymbionts: master modulators of fungal phenotypes. Microbiology Spectrum 5: e5. https://doi.org/10.1128/microb....
3.
Arenal, F., Platas, G. & Peláez, F. 2007. A new endophytic species of Preussia (Sporormiaceae) inferred from morphological observations and molecular phylogenetic analysis. Fungal Diversity 25: 1–17.
4.
Arendt, K. R., Hockett, K. L., Araldi-Brondolo, S. J., Baltrus, D. A. & Arnold, A. E. 2016. Isolation of endohyphal bacteria from foliar Ascomycota and in vitro establishment of their symbiotic associations. Applied and Environmental Microbiology 82: 2943–2949. https://doi.org/10.1128/AEM.00....
5.
Arnold, A. E. & Lutzoni, F. 2007. Diversity and host range of foliar fungal endophytes: are tropical leaves biodiversity hotspots? Ecology 88: 541–549. https://doi.org/10.1890/05-145....
6.
Arnold, A. E., Miadlikowska, J., Higgins, K. L., Sarvate, S. D., Gugger, P., Way, A., Hofstetter, V., Kauff, F. & Lutzoni, F. 2009. A phylogenetic estimation of trophic transition networks for ascomycetous fungi: are lichens cradles of symbiotrophic fungal diversification? Systematic Biology 58: 283–297. https://doi.org/10.1093/sysbio....
7.
Asgari, B. & Zare, R. 2010. Two new species of Preussia from Iran. Nova Hedwigia 90: 533–548. https://doi.org/10.1127/0029-5....
8.
Bashyal, B. P., Kithsiri Wijeratne, E. M., Faeth, S.H. & Gunatilaka, A. A. L. 2005. Globosumones A-C, cytotoxic orsellinic acid esters from the Sonoran desert endophytic fungus Chaetomium globosum. Journal of Natural Products 68: 724–728. https://doi.org/10.1021/np0580....
9.
Carbone, I. & Kohn, L. M. 1999. A method for designing primer sets for speciation studies in filamentous Ascomycetes. Mycologia 91: 553–556. https://doi.org/10.1080/002755....
10.
Crous, P. W., Cowan, D. A., Maggs-Kölling, G., Yilmaz, N., Larsson, E., et al. 2020. Fungal Planet description sheets: 1112–1181. Persoonia 45: 251–409. https://doi.org/10.3767/persoo....
11.
Crous, P. W., Cowan, D. A., Maggs-Kölling, G., Yilmaz, N., Thangavel, R., et al. 2021. Fungal Planet description sheets: 1182–1283. Persoonia 46: 313–528. https://doi.org/10.3767/persoo....
12.
Del Olmo-Ruiz, M. 2012. Diversity, distributions, and host affiliations of fungal endophytes associated with seedless vascular plants. PhD disser-tation, University of Arizona, Tucson, AZ, USA.
13.
Dimmitt, M. A. 2000. Ephedraceae (ephedra family). In: Philips, S. J. & Comus, P. W. (eds) A Natural History of the Sonoran Desert, p. 224. Arizona-Sonora Desert Museum Press.
14.
Edgar, R. C. 2004. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research 32: 1792–1797. https://doi.org/10.1093/nar/gk....
15.
Ewing, B. & Green, P. 1998. Base-calling of automated sequencer traces using phred. II. Error probabilities. Genome Research 8: 186–194.
16.
Ewing, B., Hillier, L., Wendl, M. C. & Green, P. 1998. Base-calling of automated sequencer traces using phred. Genome Research 8: 175–185.
18.
González-Juárez, D. E., Escobedo-Moratilla, A., Flores, J., Hidalgo-Figueroa, S., Martínez-Tagüeña, N., Morales-Jiménez, J., Muñiz-Ramírez, A., Pastor-Palacios, G., Pérez-Miranda, S., Ramírez-Hernández, A., Trujillo, J. & Bautista, E. 2020. A review of the Ephedra genus: distribution, ecology, ethnobotany, phytochemistry and pharmacological properties. Molecules 25: e3283. https://doi.org/10.3390/molecu....
19.
González-Menéndez, V., Martin, J., Siles, J. A., González-Tejero, M. R., Reyes, F., Platas, G., Tormo, J. R. & Genilloud, O. 2017. Biodiversity and chemotaxonomy of Preussia isolates from the Iberian Peninsula. Mycological Progress 16: 713–728. https://doi.org/10.1007/s11557....
20.
González-Menéndez, V., Crespo, G., de Pedro, N., Diaz, C., Martín, J., Serrano, R., Mackenzie, T.A., Justicia, C., González-Tejero, M. R., Casares, M., Vicente, F., Reyes, F., Tormo, J. R. & Genilloud, O. 2018. Fungal endophytes from arid areas of Andalusia: high potential sources for antifungal and antitumoral agents. Scientific Reports 8: e9729. https://doi.org/10.1038/s41598....
21.
Higgins, K. L., Arnold, A. E., Miadlikowska, J., Sarvate, S. D. & Lutzoni, F. 2007. Phylogenetic relationships, host affinity, and geographic structure of boreal and arctic endophytes from three major plant lineages. Molecular Phylogenetic and Evolution 42: 543–555. https://doi.org/10.1016/j.ympe....
22.
Hoffman, M. T. & Arnold, A. E. 2010. Diverse bacteria inhabit living hyphae of phylogenetically diverse fungal endophytes. Applied and Envi-ronmental Microbiology 76: 4063–4075. https://doi.org/10.1128/AEM.02....
23.
Huang, Y. L., Bowman, E. A., Massimo, N. C., Garber, N. P., U’Ren, J. M., Sandberg, D. C. & Arnold, A. E. 2018. Using collections data to infer biogeographic, environmental, and host structure in communities of endophytic fungi. Mycologia 110: 47–62. https://doi.org/10.1080/002755....
24.
Huelsenbeck, J. P. & Ronquist, F. 2001. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17: 754–755. https://doi.org/10.1093/bioinf....
25.
Kithsiri Wijeratne, E. M., Paranagama, P. A. & Gunatilaka, A. A. L. 2006. Five new isocoumarins from Sonoran desert plant-associated fungal strains Paraphaeosphaeria quadriseptata and Chaetomium chiversii. Tetrahedron 62: 8439–8446. https://doi.org/10.1016/j.tet.....
26.
Kornerup, A. & Wanscher, J. H. 1967. Methuen Handbook of Color. Methuen and Co., London.
27.
Kruys, A. & Wedin, M. 2009. Phylogenetic relationships and an assessment of traditionally used taxonomic characters in the Sporormiaceae (Pleosporales, Dothideomycetes, Ascomycota), utilising multi‐gene phylogenies. Systematics and Biodiversity 7: 465–478. https://doi.org/10.1017/S14772....
28.
Maddison, W. P. & Maddison, D. R. 2021. Mesquite: a modular system for evolutionary analysis. Version 3.70. http://mesquiteproject.org.
29.
Madeira, F., Pearce, M., Tivey, ARN., Basutkar, P., Lee, J., Edbali, O., Madhusoodanan, N., Kolesnikov, A. & Lopez, R. 2022. Search and sequence analysis tools services from EMBL-EBI in 2022. Nucleic Acids Research 50: w276–w279. https://doi.org/10.1093/nar/gk....
30.
Mapperson, R. R., Kotiw, M., Davis, R. A. & Dearnaley, J. D. 2014. The diversity and antimicrobial activity of Preussia sp. endophytes isolated from Australian dry rainforests. Current Microbiology 68: 30–37. https://doi.org/10.1007/s00284....
31.
Massimo, N., Devan, M. M. N., Arendt, K. R., Wilch, M., Riddle, J. M., Furr, S. H., Steen, C., U’Ren, J. M., Sandberg, D. C. & Arnold, A. E. 2015. Fungal endophytes of desert plants: infrequent in culture, but diverse and distinctive symbionts. Microbial Ecology 70: 61–76. https://doi.org/10.1007/s00248....
32.
McGinnies, W. G. 1976. An overview of the Sonoran Desert: An essay developed from a paper given at the opening session of the 2nd Annual Conference of the Consortium of Arid Lands Institutions (CALI), February 4, 1976, in Tucson, Arizona: Tucson, University of Arizona, Arid Lands Information Center. Available online: http://alic.arid.arizona.edu/s....
33.
Miller, M. A., Pfeiffer, W. & Schwartz, T. 2010. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In: Proceedings of the Gateway Computing Environments Workshop (GCE), pp. 1–8. New Orleans, LA. https://doi.org/10.1109/GCE.20....
34.
Nabhan, G. P. & Plotkin, M. J. 1994. Introduction. In: Nabhan, G. P. & Carr, J. L. (eds) Ironwood: An Ecological and Cultural Keystone of the Sonoran Desert. Conservation International, Washington, D.C.
35.
Peláez, F., Collado, J., Arenal, F., Basilio, A., Cabello, A., Díez Matas, M. T., García, J. B., González Del Val, A., González, V., Gorrochategui, J., Hernández, P., Martín, I., Platas, G. & Vicente, F. 1998. Endophytic fungi from plants living on gypsum soils as a source of secondary me-tabolites with antimicrobial activity. Mycological Research 102: 755–761. https://doi.org/10.1017/S09537....
37.
Rehner, S. A. & Buckley, E. 2005. A Beauveria phylogeny inferred from nuclear ITS and EF1-α sequences: evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 97: 84–89. https://doi.org/10.3852/mycolo....
38.
Rondeau, R. J., Van Devender, T. R., Bertelsen, C. D., Jenkins, P. D., Van Devender, R. K. & Dimmitt, M. A. 1991. Flora and vegetation of the Tucson Mountains, Pima County, Arizona. Desert Plants 12: 3–46.
39.
Shreve, F. 1951. Vegetation of the Sonoran Desert. Carnegie Institution of Washington, Publication 591.
40.
Swofford, D. L. 2003. PAUP*. Phylogenetic Analysis Using Parsimony (*and Other Methods). Version 4.0a. Sinauer Associates, Sunderland, Massachusetts.
41.
Woods, D. O. 2022. Effects of coastal processes on endophytic fungal communities. Honors thesis, University of Arizona, Tucson, AZ, USA.
42.
Zhan J. X., Liu, M. P. X., Faeth, S. H. & Gunatilaka, A. A. L. 2007. Search for cell motility and angiogenesis inhibitors with potential anticancer activity: Beauvericin and other constituents of two endophytic strains of Fusarium oxysporum. Journal of Natural Products 70: 227–232. https://doi.org/10.1021/np0603....
43.
Zwickl, D. J. 2006. Genetic algorithm approaches for the phylogenetic analysis of large biological sequence datasets under the maximum likelihood criterion [Doctoral dissertation]. The University of Texas at Austin.
CITATIONS (5):
1.
Diversity of fungi associated with petroglyph sites in the Negev Desert, Israel, and their potential role in bioweathering
Laura Rabbachin, Irit Nir, Monika Waldherr, Ylenia Vassallo, Guadalupe Piñar, Alexandra Graf, Ariel Kushmaro, Katja Sterflinger
Frontiers in Fungal Biology
Laura Rabbachin, Irit Nir, Monika Waldherr, Ylenia Vassallo, Guadalupe Piñar, Alexandra Graf, Ariel Kushmaro, Katja Sterflinger
Frontiers in Fungal Biology
2.
Preussia jejuensis
sp. nov.,
P. koreensis
sp. nov., and
P. isomera,
Coprophilous Fungi Isolated from Horse Dung in Seopjikoji, Jeju Island in the Republic of Korea
HyeongJin Noh, Hyun Uk Cho, Seong Hwan Kim
Mycobiology
HyeongJin Noh, Hyun Uk Cho, Seong Hwan Kim
Mycobiology
3.
Endophytic fungal diversity within galls induced by Asphondylia borrichiae (Diptera: Cecidomyiidae) and their surrounding host tissues (Borrichia frutescens and Iva frutescens)
Shannon L. Sharpe, Dale Casamatta, Anthony Rossi
Fungal Ecology
Shannon L. Sharpe, Dale Casamatta, Anthony Rossi
Fungal Ecology
4.
A Scytalidium-like indoor fungus revealing polyphyletic relationships and convergent evolution in Scytalidium
De-Wei Li, Ethan Paine, Neil P. Schultes
Fungal Biology
De-Wei Li, Ethan Paine, Neil P. Schultes
Fungal Biology
5.
Identity and diversity of culturable endophytic fungi associated with Capparis spinosa L. in Iran
Samira Karimzadeh, Naser Safaie, Shideh Mojerlou, Leila Ebrahimi
Scientific Reports
Samira Karimzadeh, Naser Safaie, Shideh Mojerlou, Leila Ebrahimi
Scientific Reports
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.