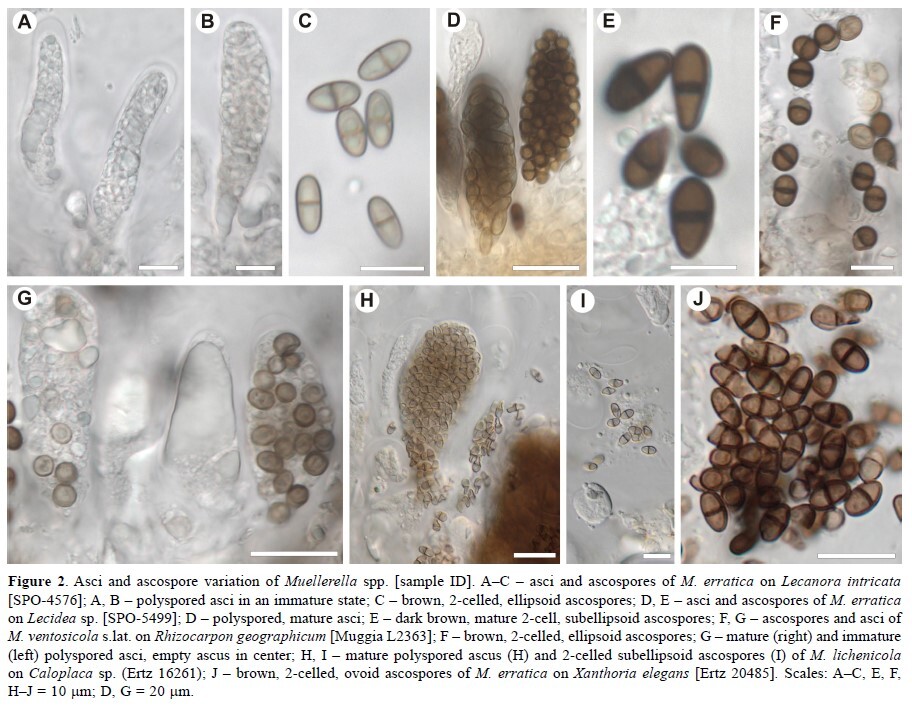
Muellerella, a lichenicolous fungal genus recovered as
polyphyletic within Chaetothyriomycetidae (Eurotiomycetes,
Ascomycota)
1
Department of Life Sciences, University of Trieste, Via Giorgieri 10,
34127 Trieste, Italy
2
Real Jardín Botánico, CSIC, Plaza de Murillo, 2, 28014 Madrid, Spain
3
Meise Botanic Garden, Department Research, Nieuwelaan 38,
B-1860 Meise, Belgium
4
Fédération Wallonie-Bruxelles, Direction Générale de l’Enseignement
non obligatoire et de la Recherche scientifique, rue A. Lavallée 1,
B-1080 Bruxelles, Belgium
Publication date: 2019-12-16
Plant and Fungal Systematics 2019; 64(2): 367-381
KEYWORDS
ABSTRACT
Molecular data and culture-dependent methods have helped to uncover the
phylogenetic relationships of numerous species of lichenicolous fungi, a specialized group
of taxa that inhabit lichens and have developed diverse degrees of specificity and parasitic
behaviors. The majority of lichenicolous fungal taxa are known in either their anamorphic or
teleomorphic states, although their anamorph-teleomorph relationships have been resolved in
only a few cases. The pycnidium-forming Lichenodiplis lecanorae and the perithecioid taxa
Muellerella atricola and M. lichenicola were recently recovered as monophyletic in Chaetothyriales
(Eurotiomycetes). Both genera are lichenicolous on multiple lichen hosts, upon
which they show a subtle morphological diversity reflected in the description of 14 species
in Muellerella (of which 12 are lichenicolous) and 12 in Lichenodiplis. Here we focus on
the teleomorphic genus Muellerella and investigate its monophyly by expanding the taxon
sampling to other species occurring on diverse lichen hosts. We generated molecular data for
two nuclear and one mitochondrial loci (28S, 18S and 16S) from environmental samples.
The present multilocus phylogeny confirms the monophyletic lineage of the teleomorphic
M. atricola and M. lichenicola with their L. lecanorae-like anamorphs, but places the rest of
the Muellerella species studied in two different monophyletic lineages with strong support.
The first, Muellerella spp. 1, is nested within some new lineages of black fungi isolated
from different epilithic lichen thalli, while the second, Muellerella spp. 2, is closely related
to the Verrucariales. Based on these results, we reappraise the phylogenetic placement of
Muellerella and suggest its polyphyly within Chaetothyriomycetidae.
REFERENCES (51)
1.
ger, P., Way, A., Hofstetter, V., Kauff, F. & Lutzoni, F. 2009. A phylogenetic estimation of trophic transition networks for ascomycetous fungi: are lichens cradles of symbiotrophic fungal diversification? Systematic Biology 58: 283–297.
2.
Aveskamp, M. M., Murace, M. A., Wounderberg, J. H. C., Groenwald, J. Z. & Crous P. W. 2009. DNA phylogeny reveals polyphyly of Phoma section Peyronellaea and multiple taxonomic novelties. Mycologia 101: 363–382.
3.
Banchi, E., Stanković, D., Fernandez-Mendoza, F., Gionechetti, F., Palavicini, A. & Muggia L. 2018. ITS2 metabarcoding analysis complements lichen mycobiome diversity data. Mycological Progress 17: 1049–1066.
4.
von Brackel, W. 2014. Kommentierter Katalog der flechtenbewohnenden Pilze Bayerns. Bibliotheca Lichenologica 109: 1–476.
5.
Crittenden, P. D., David, J. C., Hawksworth, D. L. & Campbell, F. S. 1995. Attempted isolation and success in the culturing of a broad spectrum of lichen-forming and lichenicolous fungi. New Phytologist 130: 267–297.
6.
Dettman, J. R., Jacobs, D. J. & Taylor, J. W. 2003. A multilocus genealogical approach to phylogenetic species recognition in the model eukaryote Neurospora. Evolution 57: 2703–2720.
7.
Diederich, P., Ertz, D., Lawrey, J. D., Sikaroodi, M. & Untereiner, W. A. 2013. Molecular data place the hyphomycetous lichenicolous genus Sclerococcum close to Dactylospora (Eurotiomycetes) and S. parmeliae in Cladophialophora (Chaetothyriales). Fungal Diversity 58: 61–72.
8.
Diederich, P., Lawrey, J. D. & Ertz, D. 2018. The 2018 classification and checklist of lichenicolous fungi, with 2000 nonlichenized, obligately lichenicolous taxa. The Bryologist 121: 340–425.
9.
Döbbler, P. & Triebel, D. 1985. Hepaticole Vertreter der Gattung Muellerella und Dactylospora (Ascomycetes). Botanisches Jahrbuch der Systematic 107: 503–519.
10.
Ertz, D., Diederich, P., Lawrey, J. D. & Berger, F. 2015a. Phylogenetic insights resolve Dacampiaceae (Pleosporales) as polyphyletic: Didymocyrtis (Pleosporales, Phaeosphaeriaceae) with Phoma-like anamorphs resurrected and segregated from Polycoccum (Trypetheliales, Polycoccaceae fam. nov.). Fungal Diversity 74: 53–89.
11.
Ertz, D., Lawrey, J. D., Common, R. S. & Diederich, P. 2014. Molecular data resolve a new order of Arthoniomycetes sister to the primarily lichenized Arthoniales and composed of black yeasts, lichenicolous and rock-inhabiting species. Fungal Diversity 66: 113–137.
12.
Ertz, D., Tehler, A., Irestedt, M., Frisch, A., Thor, G. & van den Boom, P. 2015b. A large-scale phylogenetic revision of Roccellaceae (Arthoniales) reveals eight new genera. Fungal Diversity 70: 31–53.
13.
Fernández-Mendoza, F., Kopun, T., Fleischhacker, A., Grube, M. & Muggia, L. 2017. ITS1 metabarcoding highlights low specificity of lichen mycobiomes at local scale. Molecular Ecology 26: 4811–4830.
14.
Fleischhacker, A., Grube, M., Kopun, T., Hafellner, J. & Muggia, L. 2015. Community analyses uncover high diversity of lichenicolous fungi in alpine habitats. Microbial Ecology 70: 348–360.
15.
Green, S. J., Venkatramanan, R. & Naqib, A. 2015. Deconstructing the Polymerase Chain Reaction: understanding and correcting bias associated with primer degeneracies and primer-template mismatches. PLoS ONE 10: e0128122. .
16.
Gueidan, C., Villaseñor, R., de Hoog, G. S., Gorbushina, A. A., Unteriener, W. A. & Lutzoni, F. 2008. A rock-inhabiting ancestor for mutualistic and pathogen-rich fungal lineages. Studies in Mycology 61: 111–119.
17.
Gueidan, C., Aptroot, A., Da Silva Cáceres, M. E., Badali, H. & Stenroos, S. 2014. A reappraisal of orders and families within the subclass Chaetothyriomycetidae (Eurotiomycetes, Ascomycota). Mycological Progress 13: 1027–1039.
18.
Hafellner, J. 2007. The lichenicolous fungi inhabiting Tephromela species. Bibliotheca Lichenologica 96: 103–128.
19.
Hall, T. A. 1999. BioEdit: a user friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acid Symposia Series 41: 95–98.
20.
Harutyunyan, S., Muggia, L. & Grube, M. 2008. Black fungi in lichens from seasonally arid habitats. Study in Mycology 61: 83–90.
21.
Hawksworth, D. L. 1979. Studies in the genus Endococcus (Ascomycotina, Dothideales). Botaniska Notiser 132: 283–290.
22.
Hawksworth, D. L. 2011. A new dawn for the naming of fungi: impacts of decisions made in Melbourne in July 2011 on the future publication and regulation of fungal names. IMA Fungus 2: 155–162.
23.
Ihlen, P. G. & Wedin, M. 2008. An annotated key to the lichenicolous Ascomycota (including mitosporic morphs) of Sweden. Nova Hedwigia 86: 275–365.
24.
Kauff, F. & Lutzoni, F. 2002. Phylogeny of the Gyalectales and Ostropales (Ascomycota, Fungi): among and within order relationships based on nuclear ribosomal RNA small and large subunits. Molecular Phylogenetic and Evolution 25: 138–156.
25.
Lawrey, J. D. & Diederich, P. 2003. Lichenicolous fungi: Interactions, evolution, and biodiversity. The Bryologist 106: 80–120.
26.
Mason-Gamer, R. J. & Kellogg, E. A. 1996. Testing for phylogenetic conflict among molecular data set in the tribe Triticeae (Gramineae). Systematic Biology 54: 524–545.
27.
Miadlikowska, J., Kauff, F., Hofstetter, V., Fraker, E., Grube, M., Hafellner, J., Reeb, V., Hodkinson, B. P., Kukwa, M. & Lücking, R., et al. 2006. New insights into classification and evolution of the Lecanoromycetes (Pezizomycotina, Ascomycota) from phylogenetic analyses of three ribosomal RNA- and two protein-coding genes. Mycologia 98: 1088–1103.
28.
Miadlikowska, J., McCune, B. & Lutzoni, F. 2002. Pseudocyphellaria perpetua, a new lichen from Western North America. The Bryologist 105: 1–10.
29.
Muggia, L. & Grube, M. 2018. Fungal diversity in lichens: from extremotolerance to interaction with algae. Life 8: 15.
30.
Muggia, L., Pérez-Ortega, S., Fryday, A., Spribille, T. & Grube, M. 2014. Global assessment of genetic variation and phenotypic plasticity in the lichen-forming species Tephromela atra. Fungal Diversity 64: 233–251.
31.
Muggia, L., Kopun, T. & Ertz, D. 2015. Phylogenetic placement of the lichenicolous, anamorphic genus Lichenodiplis and its connection to Muellerella-like teleomorphs. Fungal Biology 119: 1115–1128.
32.
Muggia, L., Fleischhacker, A., Kopun, T. & Grube, M. 2016. Extremotolerant fungi from alpine rock lichens and their phylogenetic relationships. Fungal Diversity 76: 119–142.
33.
Muggia, L., Kopun, T. & Grube, M. 2017. Effects of growth media on the diversity of culturable fungi from lichens. Molecules 22: 824.
34.
Pérez-Ortega, S., Suija, A. & de los Ríos, A. 2011.The connection between Abrothallus and its anamorph state Vouauxiomyces established by Denaturing Gradient Gel Electrophoresis (DGGE). The Lichenologist 43: 277–279.
35.
Pino-Bodas, R., Zhurbenko, M. P. & Stenroos, S. 2017. Phylogenetic placement within Lecanoromycetes of lichenicolous fungi associated with Cladonia and some other genera. Persoonia 39: 91–117.
36.
Rai, M. K., Vaibhav, V., Tiwari, V. V., Irinyi, L. & Kövics, G. J. 2014. Advances in taxonomy of genus Phoma: polyphyletic nature and role of phenotypic traits and molecular systematics. Indian Journal of Microbiology 54: 123–128.
37.
Reeb, V., Lutzoni, F. & Roux, C. 2004. Contribution of RPB2 to multilocus phylogenetic studies of the euascomycetes (Pezizomycotina, Fungi) with special emphasis on the lichen-forming Acarosporaceae and evolution of polyspory. Molecular Phylogenetic and Evolution 32: 1036–1060.
38.
Ruibal, C., Gueidan, C., Selbmann, L., Gorbushina, A. A., Crous, P. W., Groenewald, J. Z., Muggia, L., Grube, M., Isola, D., Schoch, C. L., Staley, J. T., Lutzoni, F. & de Hoog, G. S. 2009. Phylogeny of rock-inhabiting fungi related to Dothideomycetes. Study in Mycology 64: 123–133.
39.
Selbmann, L., Grube, M., Onofri, S., Isola, D. & Zucconi, L. 2013. Antarctic epilithic lichens as niches for black meristematic fungi. Biology 2: 784–797.
40.
Stamatakis, A. 2014. RAxML Verison.8.2: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics, open access link: http://bioinformatics.oxfordjo...? keytype=ref&ijkey=VTEqgUJYCDcf0kP.
41.
Tedersoo, L., Sanchez-Ramırez, S., Koljalg, V., Bahram, V., Döring, M., Schigel, D., May, T., Ryberg, M. & Abarenkov, K. 2018. High-level classification of the Fungi and a tool for evolutionary ecological analyses. Fungal Diversity 90: 135–159.
42.
Teixeira, M. M., Moreno, L. F., Stielow, B. J., Muszewska, A., Hainaut, M., Gonzaga, L. & Abouelleil A., et al., 2017. Exploring the genomic diversity of black yeasts and relatives (Chaetothyriales, Ascomycota). Studies in Mycology 86: 1–28.
43.
Triebel, D. 1989. Lecideicole Ascomyceten . Eine Revision der obligat lichenicolen Ascomyceten auf lecideoiden Flechten. Bibliotheca Lichenologica 35: 1–278.
44.
Triebel, D. & Kainz, C. 2004. Muellerella. In: Nash, T. H., Ryan, B. D., Diederich, P., Gries, C. & Bungartz, F. (eds), Lichen Flora of the Greater Sonoran Desert Region, Vol. 2., pp. 673–675. Lichens Unlimited, Arizona State University, Tempe, Arizona.
45.
U’Ren, J. M., Lutzoni, F. M., Miadlikowska, J. & Arnold, A. E. 2010. Community analysis reveals close affinities between endophytic and endolichenic fungi in mosses and lichens. Microbial Ecology 60: 340–53.
46.
U’Ren, J. M., Lutzoni, F., Miadlikowska, J., Laetsch, A. D. & Arnold, A. E. 2012. Host and geographic structure of endophytic and endolichenic fungi at a continental scale. American Journal of Botany 99: 898–914.
47.
U’Ren, J. M., Riddle, J. M. & Monacell, J. T. et al. 2014. Tissue storage and primer selection influence pyrosequencing-based inferences of diversity and community composition of endolichenic and endophytic fungi. Molecular Ecology Resources 14: 1032–1048.
48.
Vasse, M., Voglmayr, H., Mayer, V., Gueidan, C., Nepel, M., Moreno, L., de Hoog, S. G., Selosse, M. A., McKey, D. & Blatrix, R. 2017. A phylogenetic perspective on the association between ants (Hymenoptera: Formicidae) and black yeasts (Ascomycota: Chaetothyriales). Proceedings of the Royal Society B 284: 20162519.
49.
Vilgalys, R. & Hester, M. 1990. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. Journal of Bacteriology 172: 4238–4246.
50.
Zhou, S. & Stanosz, G. R. 2001. Primers for amplification of mtSSU rDNA, and a phylogenetic study of Botryosphaeria and associated anamorphic fungi. Mycological Research 105: 1033–1044.
51.
Zoller, S., Scheidegger, C. & Sperisen, C. 1999. PCR primers for the amplification of mitochondrial small subunit ribosomal DNA of lichen-forming ascomycetes. The Lichenologist 31: 511–516.
CITATIONS (14):
1.
Contributions to the Bulgarian lichenicolous mycota – an annotated checklist and new records
Veselin Shivarov, Nóra Varga, László Lőkös, Brackel von, Anna Ganeva, Rayna Natcheva, Edit Farkas
Herzogia
Veselin Shivarov, Nóra Varga, László Lőkös, Brackel von, Anna Ganeva, Rayna Natcheva, Edit Farkas
Herzogia
2.
Sequence data from isolated lichen-associated melanized fungi enhance delimitation of two new lineages within Chaetothyriomycetidae
Lucia Muggia, Yu Quan, Cécile Gueidan, Abdullah Al-Hatmi, Martin Grube, Hoog de
Mycological Progress
Lucia Muggia, Yu Quan, Cécile Gueidan, Abdullah Al-Hatmi, Martin Grube, Hoog de
Mycological Progress
3.
The yeast lichenosphere: High diversity of basidiomycetes from the lichens Tephromela atra and Rhizoplaca melanophthalma
Agnese Cometto, Steven Leavitt, Ana Millanes, Mats Wedin, Martin Grube, Lucia Muggia
Fungal Biology
Agnese Cometto, Steven Leavitt, Ana Millanes, Mats Wedin, Martin Grube, Lucia Muggia
Fungal Biology
4.
The origin of human pathogenicity and biological interactions in Chaetothyriales
Yu Quan, Shuwen Deng, Francesc Prenafeta-Boldủ, Veronika Mayer, Lucia Muggia, Agnese Cometto, Vania Vicente, Silva da, Maria Grisolia, Yinggai Song, Sarah Ahmed, Xueke Niu, Souza de, Peiying Feng, Roxana Vitale, Marcus Teixeira, Montarop Sudhadham, Azevedo de, Anamelia Bocca, Gerhard Haase, Laura Selbmann, Dongmei Shi, Yingqian Kang, Hoog de
Fungal Diversity
Yu Quan, Shuwen Deng, Francesc Prenafeta-Boldủ, Veronika Mayer, Lucia Muggia, Agnese Cometto, Vania Vicente, Silva da, Maria Grisolia, Yinggai Song, Sarah Ahmed, Xueke Niu, Souza de, Peiying Feng, Roxana Vitale, Marcus Teixeira, Montarop Sudhadham, Azevedo de, Anamelia Bocca, Gerhard Haase, Laura Selbmann, Dongmei Shi, Yingqian Kang, Hoog de
Fungal Diversity
5.
Tackling fungal diversity in lichen symbioses: molecular and morphological data recognize new lineages in Chaetothyriales (Eurotiomycetes, Ascomycota)
Agnese Cometto, Steven Leavitt, Martin Grube, Hoog de, LUCIA MUGGIA
Agnese Cometto, Steven Leavitt, Martin Grube, Hoog de, LUCIA MUGGIA
6.
Tackling fungal diversity in lichen symbioses: molecular and morphological data recognize new lineages in Chaetothyriales (Eurotiomycetes, Ascomycota)
Agnese Cometto, Steven Leavitt, Martin Grube, Hoog De, Lucia Muggia
Mycological Progress
Agnese Cometto, Steven Leavitt, Martin Grube, Hoog De, Lucia Muggia
Mycological Progress
7.
First molecular evidence of lichen-inhabiting
Acrospermum
and new insights into the evolution of lifestyles of Acrospermales (Dothideomycetes)
Valerii Darmostuk, Adam Flakus
Mycologia
Valerii Darmostuk, Adam Flakus
Mycologia
8.
Highly heterogeneous mycobiota shape fungal diversity in two globally distributed lichens
Agnese Cometto, Claudio G. Ametrano, Roberto De Carolis, Steven D. Leavitt, Martin Grube, Alberto Pallavicini, Lucia Muggia
Fungal Ecology
Agnese Cometto, Claudio G. Ametrano, Roberto De Carolis, Steven D. Leavitt, Martin Grube, Alberto Pallavicini, Lucia Muggia
Fungal Ecology
9.
Genomic features of lichen‐associated black fungi
Victoria Keller, Anjuli Calchera, Jürgen Otte, Imke Schmitt
IUBMB Life
Victoria Keller, Anjuli Calchera, Jürgen Otte, Imke Schmitt
IUBMB Life
10.
In silico search reveals the association of lichens with black yeast-like fungi in the order Chaetothyriales
Flavia de Fatima Costa, Bruno Paulo Rodrigues Lustosa, Camila Pereira Perico, Ricardo Belmonte-Lopes, João Lucas Vitório Ribeiro Carvalho, Emanuel L. Razzolini, Germana Davila dos Santos, Bruna Jacomel Favoreto de Souza Lima, Cristina Maria de Souza-Motta, Roberto Tadeu Raittz, Yinggai Song, Laura Selbmann, G. Sybren de Hoog, Jacques Meis, Vania Aparecida Vicente
Fungal Biology
Flavia de Fatima Costa, Bruno Paulo Rodrigues Lustosa, Camila Pereira Perico, Ricardo Belmonte-Lopes, João Lucas Vitório Ribeiro Carvalho, Emanuel L. Razzolini, Germana Davila dos Santos, Bruna Jacomel Favoreto de Souza Lima, Cristina Maria de Souza-Motta, Roberto Tadeu Raittz, Yinggai Song, Laura Selbmann, G. Sybren de Hoog, Jacques Meis, Vania Aparecida Vicente
Fungal Biology
11.
The 2025 classification and checklist of lichenicolous fungi: documenting a rapidly growing knowledge of diversity
Paul Diederich, James D. Lawrey, Damien Ertz
The Bryologist
Paul Diederich, James D. Lawrey, Damien Ertz
The Bryologist
12.
Multispored Pyrenocarpous Lichenicolous Fungi of India Including Muellerella Aspiciliae sp. Nova
Y. Joshi, S. Bisht, P. Bansal, N. Pargaien
Acta Botanica Hungarica
Y. Joshi, S. Bisht, P. Bansal, N. Pargaien
Acta Botanica Hungarica
13.
Ubiquitous occurrence of the black fungus Melanina gundecimermaniae in the lichen Umbilicaria pustulata
Victoria Keller, Anjuli Calchera, Jürgen Otte, Veera Tuovinen Nogerius, Imke Schmitt
Current Biology
Victoria Keller, Anjuli Calchera, Jürgen Otte, Veera Tuovinen Nogerius, Imke Schmitt
Current Biology
14.
Mycobiomes of Six Lichen Species from the Russian Subarctic: A Culture-Independent Analysis and Cultivation Study
Armen Hakobjanyan, Alexey Melekhin, Marina Sukhacheva, Alexey Beletsky, Timofey Pankratov
Journal of Fungi
Armen Hakobjanyan, Alexey Melekhin, Marina Sukhacheva, Alexey Beletsky, Timofey Pankratov
Journal of Fungi
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.